Adenine transversion editors enable precise, efficient A•T-to-C•G base editing in mammalian cells and embryos
腺嘌呤碱基编辑器;A•T-to-C•G
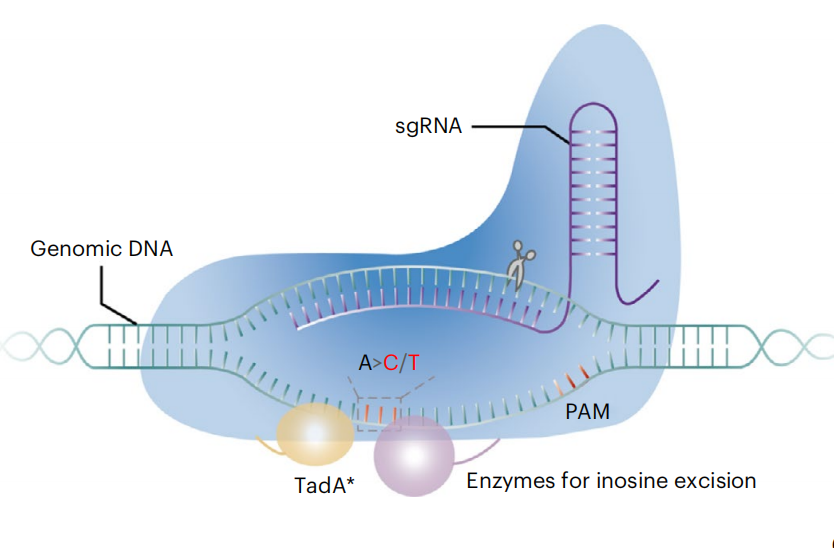
Human genetic disorders are mainly caused by genetic variants, ofwhich approximately half are pathogenic single-nucleotide variations(SNVs)1 . Precise DNA sequence conversions can be achieved throughgene editing technologies, such as programmable nuclease-inducedhomology-directed repair (HDR), base editing and prime editing2 .HDR is a double-strand DNA break (DSB)-dependent process that isinefficient in most therapeutically relevant cell types and requiresDNA donor templates. Moreover, DSBs are associated with undesiredside effects, such as P53 activation, large DNA fragment deletions andchromosomal abnormalities, such as translocations3 . Prime editorsare highly versatile and precise tools for genome editing, but their application can be limited by the need to test a variety of designs to achieveefficient editing, especially in primary cells and in vivo4–6, although recently described engineered prime editing guide RNA (epegRNA)7and PE4/5max prime editing systems8 have greatly improved prime editing efficiencies in many living systems.For some single-nucleotide conversions, base editing remains the most efficient technology to install base substitutions without inducing DSBs or requiring donor templates. Cytosine base editors (CBEs) efficiently generate C•G-to-T•A conversions and are composed of a Cas9 nickase, cytosine deaminase and uracil glycosylase inhibitor (UGI) to inhibit cytosine transversion side products, including C•G-to-G•C and C•G-to-A•T, which are induced by the uracil DNA N-glycosylase (UNG)-mediated base excision repair (BER) pathway9,10. By substituting the CBE UGI with UNG or DNA repair factors, several C•G-to-G•C base editors (CGBEs) that increase the frequency of cytosine transversion editing were developed that mainly induce C-to-G conversions in mammalian cells but C-to-A transversion in Escherichia coli9–13. Adenine base editors (ABEs), composed of Cas9 nickase and laboratory-evolved TadA deaminases, create A•T-to-G•C conversions with few byproducts. The product purity of ABEs can be very high (≥99% from ABE7.10 at some targets, for example), likely due to a lack of efficient or well-expressed endogenous DNA glycosylases that initiate BER at inosines to induce insertions and deletions (indels) and adenine transversions14.Adenine transversions could play an important role in therapeutic gene correction and other applications for genome engineering. The ability to make targeted A•T-to-C•G and A•T-to-T•A substitutions could potentially correct 17% and 8% of known pathogenic SNVs, respectively (Fig. 1a). To date, however, no adenine transversion base editors have been described14,15. In addition to corrective gene editing, programmable adenine transversions would increase the diversity of outcomes for mutagenesis-based applications, such as lineage tracing, genetic screening and molecular evolution. Here we describe the development of adenine transversion base editors consisting of a Cas9 nickase, the highly active evolved deoxyadenine deaminase TadA-8e and mouse alkyladenine/3-methyladenine DNA glycosylase (mAAG) variants. Through the laboratory evolution of mAAG and embedding of the mAAG and TadA-8e in Cas9 nickase, A•T-to-C•G base editors (ACBEs) were developed that generate efficient and precise A•T-to-C•G conversion in mammalian cells. ACBEs can install or correct pathogenic SNVs in mouse embryos or in human cells. The development of adenine base transversion editors substantially expands the capabilities and application scope of base editing.
---------------


